A Decade of Decapods
June, 2024
Later this year, Crab Team will round our 10 year anniversary, having smashed the champagne bottle over our first Fukui trap in August 2015 (August 14 at 11:30 am at the Penn Cove site, if we’re being precise). This year gives us an incredible opportunity to both reflect and celebrate. It’s become part of the Crab Team origin story that we didn’t expect to find green crab for a much longer time, but the timing proved to be fortuitous.

OG Penn Cove monitors Dave and Susan, who are still on the team today, pull their first Fukui in 2015. Photo courtesy of Deb Paros (also OG).
The program has also been incredibly lucky to still be working with some of the same original volunteers that dove into the mud – waders first, thankfully – with us that August. These folks have been the definition of dedicated, becoming the hands down experts on their sites and the decapods that live there.
From the original 7 sites, Crab Team now numbers 68 monitoring sites from Willapa Bay to Drayton Harbor. Over time, the data itself, collected consistently, in some cases by the same individuals every month, has also accrued value. So-called long term ecological datasets are rare and valuable in learning about how ecosystems respond to long term changes and help distinguish the signal of a directional change, say from green crab invasion, relative to the noise of decadal climate cycles, and development or restoration of sites.
Biological invasions often play out on a timescale longer than the news cycle, and managing them requires sustained effort and a good long term memory. So, we’re exceedingly grateful for the commitment of community members who have helped keep the traps baited and fishing for the last 10 years and those who will sustain us into the future. Even 10 years in, we are still eager to welcome new crabbers on board! Come celebrate the summer solstice with us in the Sound-wide Molt Blitz on June 21.
All our pinchy best,
Crab Team HQ 🦀
Green Crab Migration and Movement
One of the most pressing issues for management of European green crabs in the Pacific Northwest is to better understand how green crabs, as adults, move across the water bodies they inhabit. We are increasingly learning about the oceanographic influence of tides, currents, and storms on how larvae are transported into and around Washington shorelines (Du et al. 2024). Even though adult crabs are thought to move shorter distances, knowing adult movement patterns is also critical, enabling more effective trapping, and hopefully reducing population sizes. Some of the basic questions that we have are:
- How far do green crabs travel on a given tide or series of tides?
- Do they use different tidal elevations or habitat types at different times of year?
- Do males and females move differently, or target different habitat types?
- Are there microhabitat types (e.g, types of vegetation or other cover) that green crabs spend time in that we aren’t trapping them in?
Answers to these questions apply directly to strategies for green crab control efforts and tell us, for instance, how large of an area a single trap might attract green crabs from. They could also suggest where we place traps at different times of year to catch the most crabs, or how we interpret observed catch rates based on where, when, and how traps were set.
What we Know from Trapping Observations
We have – at least in part – some insight to these questions. The most readily available information is just a sense of where we find crabs by trapping and when. Trappers have long observed that green crabs are, compared to the warmer months, less conspicuous during winter and much harder to find and trap; they become less active and often hunker down for long periods. As a result they are less likely to be captured in baited traps that are frequently set in intertidal areas (their preferred habitat during warmer months). In areas where green crab numbers are low this can give the impression that they’ve all but disappeared. Little is known about where they go and what they do in the colder, darker months of the year. Until recently, most wintertime observations of green crabs came from shellfish growers, who sometimes encountered crabs nestled among their gear in shallow subtidal areas. The first detections in Samish Bay were made this way, and reports from growers in Willapa Bay in 2016 and 2017 occurred during the winter. As waters warm and day length increases, green crabs start to show up in greater numbers in intertidal traps in the spring.
Movement Studies
This information isn’t perfect. Trapping only tells you one location that the crabs are using – the place you set the traps! There’s always the possibility, and the fear, that crabs are spending most of their time somewhere else that you aren’t looking. So why not ask the crabs to tell us where they are hiding out?
One way to track movement of green crabs over longer periods is a mark-release-recapture study. Crabs are individually tagged and released, and if they are found again, most often in another trap set, we can learn the crab’s position at at least two time points. If the crab is released again (and falls for the bait again), we can gather additional time points and locations for individual crabs, ultimately resulting in a better picture of the range and movement of an individual crab, as opposed to the location of a population.
Mark-release-recapture studies can be technically and logistically challenging, and with an invasive species, the idea of releasing (and re-releasing) a crab back into the environment is not always easy to swallow, or even allowed. Other challenges include loss of the tags, especially due to molting, and the fact that recaptures are somewhat rare, meaning a LOT of crabs have to be tagged, and a LOT of traps have to be set to be able to get good information. One group currently working on tracking crabs this way is the Makah Fisheries department. For the past two years, the Makah team has been using this type of study to understand how green crabs in two river systems of Makah Bay (the Wa’atch and Tsoo-Yess) are using the rivers and adjacent beach habitat.
Early Forays into Crab Telemetry

Sean McDonald submerges the directional hydrophone to listen for pings from tagged green crabs at Tokeland, WA. (Photo courtesy of Sean McDonald)
Another method to track animal movement is acoustic telemetry. Like the radio collars and tags used to track elk, wolves, lions, and even bees, crabs can be affixed with acoustic pingers. Crab Team co-PI Sean McDonald used active acoustic telemetry to track European green crabs in California (July, 2001) and Washington (July 2002). At the time, the tags were relatively small, about the size of a pen cap, battery-powered devices that emitted a coded acoustic signal (or “ping”) that could be detected with a directional hydrophone. By triangulating on these unique signals, the research team could accurately determine a crab’s location. Re-locating the crabs at regular intervals enables researchers to establish the position of the crab at multiple time points, similar to recaptures in mark-release-recapture studies, but often with a higher “re-sighting” rate since the researchers go out to find the crabs. In Sean’s case, these re-sight efforts took place four times a day, about every six hours or so – yes, even snorkeling at night, in the dark – for two weeks! “It was painstaking work but it allowed us to acquire very precise information on movements and the habitats green crabs occupy.”
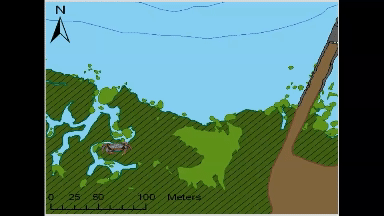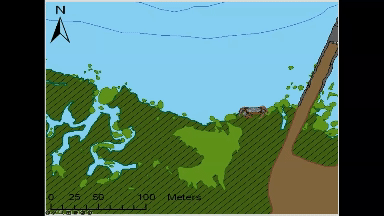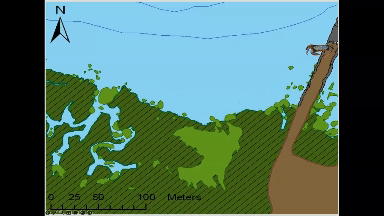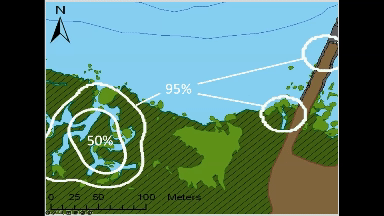
Animated GIF of movement of an individual green crab tagged and tracked at Kindred Slough in Tokeland as part of Sean McDonald’s research. The white circles at the end of the looped animation show the areas where the crab spent 50% and 95% of the total tracked time over two weeks.
Most crabs in this study (9 each from both locations) stayed within a few hundred meters of where researchers tagged and released them, often opting to hunker down among rocks or vegetation rather than venturing long distances. The crabs tagged near Tokeland, for instance, tended to move laterally along the shoreline and around the marina jetty, frequently utilizing native marsh vegetation, patches of cordgrass, or rip-rap. Rather than undertaking lengthy migrations between deeper water refuges and higher intertidal feeding grounds, as had been observed in Dungeness crabs in other studies (Holsman et al. 2006), green crabs, on average, remained in an area not much larger than a basketball court.
Time and Tides Bring Better Tech
Early telemetry data added to our understanding of green crab behavior, and confirmed some of the conjectures from trapping observations, but they were quite limited. Efforts were restricted by both battery life (about two weeks) and researcher endurance (also about two weeks). Seasonal migratory patterns of green crab are likely also important, but crabs would need to be tracked for much longer than two weeks to be able to directly observe where they go in the cooler months.

Green crab with acoustic tag used by Curtis Roegner in recent tracking studies. Photo courtesy of C. Roegner
Luckily, recent technological advancements in telemetry gear and battery life have created new and affordable tools that are beginning to shed some light on the hidden world of green crabs and researchers in several regions are taking a peek. A research team based in Maine recently used passive acoustic telemetry to investigate winter movements in the Webhannet River Estuary (Zarrella-Smith et al. 2022). Similar to Sean’s research, the team affixed “pingers” to a number of crabs and released them back into the estuary. However, rather than chasing down the crabs with a hydrophone, they were able to use a series of automated receivers positioned throughout the estuary, and listen in on the crabs’ pings remotely. Thanks to improved battery life, and lower impact on researchers, the team tracked 22 crabs in this way for nearly a year (July 2018 through April 2019).
Much like our earlier observations from the West Coast, it turns out these crabs stuck to their own little areas of the estuary, with each area being about 300-600 m (or roughly 3-6 American football fields) in length. But when the temperature dropped below 10°C, the crabs generally moved downstream. In the winter, nine of the crabs were found to be “overwintering” in the deeper waters of the lower estuary, likely to avoid the cold.
Closer to home, Dr. Curtis Roegner and other researchers from NOAA’s Northwest Fisheries Science Center set out to conduct a similar study in Willapa Bay. In September of 2022, Dr. Roegner and his team (shout out to Zach Forster and David Beugli who are both Crab Team monitors!) deployed two arrays of passive acoustic receivers in the vicinity of Nahcotta and tagged forty green crabs. Their goal was to better understand what green crabs do during winter. They retrieved the data at the end of February, 2023 (after 139 days of tracking) and began processing it. The early results are providing additional evidence of seasonal activity, or rather, seasonal inactivity.
“There were a few localized movements…”, said Dr. Roegner, “but most stayed put. In contrast, the majority of the sixteen Dungeness [crabs] we tagged moved away [from the release location and array]”.
Roegner’s team noted that more than half of the tagged green crab remained at the release site for the entire observation period, whereas all of the Dungeness crabs left the area by about day 14 (Roegner et al. 2023). The winter study also showed that green crabs do use shallow subtidal habitats during these colder months, but are still in the intertidal about 30% of the time. This is in agreement with observations by shellfish growers that can find them in their deeper gear in colder weather. By contrast, Dungeness crabs were almost strictly observed in subtidal habitats. Similar to the way trapping captures more green crabs around protective habitat structure, Roegner’s telemetry confirmed that green crabs did spend more time around oyster bags and reefs. Lastly, though the majority of travel distances were small, less than 300 m, some long forays did occur, indicating that in winter, green crabs can, on occasion, migrate out of a site, and to another.
The Next Move is Ours
These studies continue to improve our resolution of the highly seasonal movements and activity patterns of green crabs. Green crabs appear to predominantly occupy high intertidal habitat (greater than 0 feet above Mean Lower Low Water; MLLW) throughout much of the spring and summer. During this period, the crabs occupy a variety of structured habitat, like shell and marsh vegetation, for shelter and show a particular affinity for shallow sloughs, pools, and jetties, and most of the time they don’t travel very far. Once the weather cools in autumn and the days grow shorter, activity levels decline and the crabs may move into slightly deeper water to take refuge in the shallow subtidal zone.
These studies have confirmed some of the patterns observed in trapping efforts. By knowing where green crabs are spending more of their time, and when, trappers can target their gear to match seasonal movement. It would make sense, for instance, to focus trapping at intertidal elevations in the summer, but shift traps slightly deeper in the winter to account for “the hunker factor.”
-P. Sean McDonald and Emily Grason
Header image: Green crab with a slightly older version of acoustic tags used in early 2000’s California and Washington studies. Photo: P. Sean McDonald.
Featured Creature: Salmonids
Common Name and Species Names:
- Chinook salmon (Oncorhynchus tshawytscha)
- Chum salmon (Oncorhynchus keta)
- Sockeye salmon (Oncorhynchus nerka)
- Pink salmon (Oncorhynchus gorbuscha)
- Coho salmon (Oncorhynchus kisutch)
- Steelhead trout (Oncorhynchus mykiss)
- Cutthroat trout (Oncorhynchus clarkii)
Creature Feature: Salmonids
Let’s talk about salmonids! We know ‘em, love ‘em, and maybe even love to eat ‘em but why are we dedicating a whole Creature Feature to ‘em?
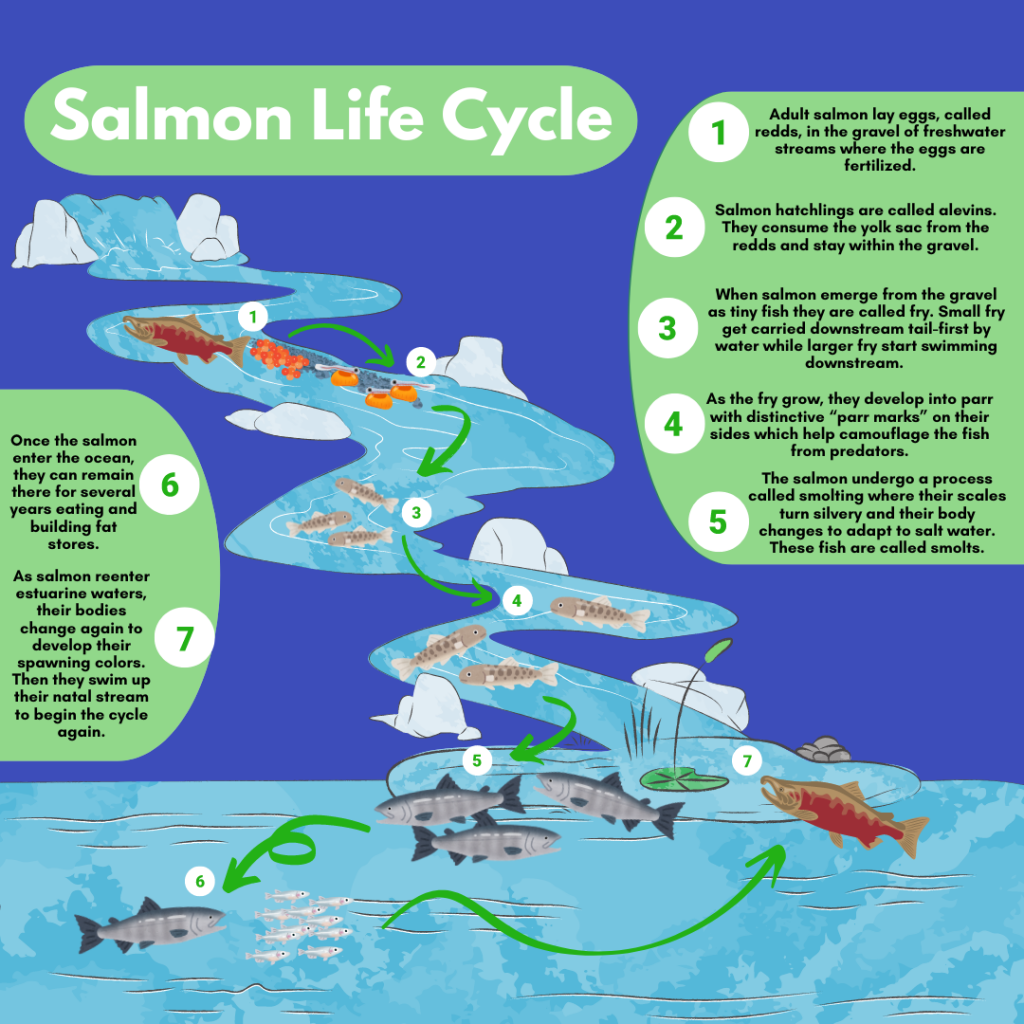
Salmon spend different parts of their lives migrating from stream to ocean and back again, bringing them through estuarine sites that green crabs favor more than once (5 and 7).
Salmonids, including salmon and trout, are anadromous fishes, meaning they spend the early part of their life cycle in freshwater and most of their adult life in the open ocean before returning to their home stream to spawn and die. These fishes are culturally and economically important to our region and their migrations send them right through prime green crab habitat. This means that European green crabs could have potential impacts on these species, and also that there is a chance, if small, Crab Team monitors could encounter them while on site.
Most of the impacts we talk about with green crab are “direct,” the result of green crabs eating or competing with another species. But the impacts to salmon are more likely to be “indirect” – green crabs might harm salmonid populations by damaging habitats where they overlap. The pocket estuaries, salt marshes, and tide flats, where many Crab Team sites are monitored, tend to have little wave action, freshwater input from a surface stream, and are also tidally influenced. These factors combined provide fantastic habitat for juvenile salmonids as well as the forage fish that are important food for salmon as they grow larger. Unfortunately, European green crabs have been found to damage habitats through their burrowing and foraging behaviors. Green crabs can disturb eelgrass meadows by disrupting sediment or damaging eelgrass roots or runners while foraging for clams and other invertebrates. The loosening of sediments can expose or weaken eelgrass roots which then can be uprooted from wave action or currents. Green crabs have also been found to eat eelgrass, and while this is not their preferred source of food it can do in a pinch when green crab densities are high and there is increased competition for prey. Take a peek at our 2021 newsletter article for more information on the habitat impacts of green crabs!
Clearing the Way for Salmonid Recovery
Salmon once lived in any river or stream that was not blocked by natural barriers. Today, salmonids face many obstacles in their journeys back and forth to and from the ocean. Whether traversing narrow culverts under roads or climbing tricky fish ladders, these fish are constantly fighting against man-made structures just to fulfill their lifecycle. There are an estimated 18,000 barriers that either partially or fully block salmonids from reaching spawning grounds in Washington state alone. Most of these barriers occur where roads cross streams and flow is restricted to pipes called culverts. If these culverts are poorly designed, they prevent salmon from swimming upstream, severely restricting spawning habitat. Other well known examples are large dams that not only block salmon passage upstream to spawning grounds, but also moderate river flow at the mouth, reducing the extent of delta marshes that offer shelter to migrating salmon.
Many initiatives are under way to reverse the dramatic declines of salmon caused by barriers and other factors. One important tool that protects salmon is the listing of 28 populations groups of salmon and steelhead as threatened or endangered under the Endangered Species Act (ESA).
Undoubtedly, tribes have been leaders in many efforts to protect and restore salmon populations, creating and using legislative protections for habitat and fish, and advocating for and implementing critical restoration efforts. Salmon are of existential importance to many tribes in Washington, and tribes retained the right to harvest fish in their “usual and accustomed” places when making treaties. The landmark court case that protects these rights, the Boldt Decision, ruled 50 years ago that tribes are entitled to half the salmon catch in the state. This decision, and the ESA listing of salmon populations are often the basis for the large scale of restoration efforts experts say is needed to stop the decline of salmon populations. Northwest Treaty Tribes sued the state of Washington in 2001 on the grounds that fish-blocking culverts reduced their treaty rights to harvest fish and since 2005, more than 3,700 barriers have been corrected. Yet, more funding is needed to meet the court-ordered deadline of correcting all identified barriers by 2030. The Lower Elwha Tribe was instrumental in advocating for the removal of the two Elwha dams, a process that started a decade ago and is seeing the return of coho harvest for the first time in 2023. The Stillaguamish Tribe has been partnering with WDFW to restore critical delta habitat at Leque Island and zis a ba.
Another type of restoration is improving nearshore vegetative buffers around key salmonid habitats. In Washington, it is estimated that 50-90% of areas along waterways have been extensively modified by human activity. These areas are important for salmonids as the vegetation provides shade from the sun that cools water and filters runoff providing cool, clean water for fish. A healthy nearshore vegetative buffer also contributes to habitat heterogeneity by dropping branches into the water which provides quality habitat for salmonid spawning or for refuge from predators.
One example of the importance of this kind of restoration work is the Jimmycomelately Creek and Estuary restoration led by the Jamestown S’Klallam Tribe. In 1999, only seven adult summer chum salmon returned to Jimmycomelately. Seeing the dire need of this population, work began in 2002 with channel realignment which moved the creek back to one of its historical channels which was first changed in the early 1900’s to create space for farming. The tribe also replaced the Highway 101 bridge that stretched over the creek to allow for better fish passage and accommodate flood flows. Later efforts focused on riparian and floodplain vegetation establishment and estuary restoration. All of this hard work has paid off and now Jimmycomelately Creek has had an average return of nearly 3,000 summer chum each year. Check out the Northwest Treaty Tribes website to learn more about projects related to habitat improvement that are underway in the region.
Crab Team Monitoring Efforts at Restoration Sites

An interpretive sign showing the restoration of Cypress Island (site #541) overlooking the salt marsh. Photo credit: Emily Grason
Salmon restoration efforts quite often create and focus on nearshore habitat types that are also highly suitable for European green crab, so it’s no coincidence that more than a few restoration sites are also Crab Team monitoring sites. Some, like Jimmycomelately Creek (Site #199), and Cypress Island (Site #541), are “post-restoration,” that is, Crab Team didn’t start monitoring until after the restoration efforts took place. Others, like the Duckabush on Hood Canal (Site #138) and Titlow Lagoon in Tacoma (Site #299) are slated for restoration projects in the future.
The data monitors collect at these sites offer not only the chance to keep an eye out for green crab, but also a unique opportunity to improve the understanding of how restoration might benefit other species. Most restoration projects have limited budget for monitoring, meaning it might not last more than a year or two post restoration, and might only focus on salmonids, overlooking all the other native organisms that benefit from restoring natural shoreline features and flow. Crab Team fills the gap by tracking the shore crabs, and the sculpins, and the dungies as they return.

By keeping waterways clean from litter and pollutants, improving vegetative buffers around habitats and improving fish passage there are several ways we can be stewards to the habitats that salmonids depend on!
The overlap in habitat also means that across the monitoring network there are a few sites where ESA-listed salmonid species might encounter Crab Team traps. The species/populations include Puget Sound Chinook salmon (Oncorhynchus tshawytscha), Hood Canal summer-run Chum salmon (O. keta), Ozette Lake Sockeye salmon (O. nerka) and Puget Sound Steelhead trout (O. mykiss). Thankfully, it’s extraordinarily rare for any salmonid to enter a green crab trap, and in 10 years of sampling, 15,351 trap sets, and more than 700,000 organisms trapped, Crab Team has never yet captured an ESA-listed salmonid. The few salmonids that have made an appearance include juvenile coho salmon (O. kisutch) heading out to adventure in the open ocean, and a couple coastal cutthroat (O. clarkii) on their trip back to their natal streams. While the risk of trapping salmonids as bycatch is low, it’s not impossible! Because of this, Crab Team has a five-year ESA permit that allows incidental take of these species. Monitors are all trained to look for and treat all salmonids and forage fish with extra care and attention, to ensure we can document any catch accurately and reduce the chance of harming them.
The significance of salmonids extends far beyond its role as a delicious food source. While these species rely on a variety of habitats, the presence of these fishes influences the health of entire ecosystems and are an essential part of the many cultures and economies of the Pacific Northwest. By taking special care of salmonids if they show up in Crab Team traps and by doing diligent monthly estuary monitoring, we can both protect salmonids and their habitats.
– Elyse Kelsey

If you asked many joggers and cyclists in the Anacortes area where to go for a beautiful jaunt, they’d probably point you to the Tommy Thompson Trail. It crosses shallow Fidalgo Bay along the top of an old railroad trestle. If you timed it right, under the eastern terminus of that Bay-bisecting trestle, you’d find volunteers hard at work.
Crab Team established a monitoring site at Fidalgo Trestle in 2017, which continues to be supported 7 years later by Paul and Erik Dinnel and Barry and Laurie Rotner, all original monitors who have stuck around since the start. Like many Crab Team sites, the experience for this team has included “good work and fun and lots of critters”, with treasured memories formed “in the park-and-ride lot pulling boots on and catching up”, according to Pete Haase, the fifth member of that original team, who continues to be a supporter even though he has retired his waders.
Crabs at the Trestle
What the team captures at the trestle site is a bit unusual for a Crab Team survey. Monitors consistently report a large number of cancrid crabs, most commonly graceful, as well as helmet crabs in their traps. Even once a lemon peel nudibranch! However, this site rounds out the geographic and habitat range covered by the three monitoring sites in Fidalgo Bay – the others being Crandall Spit and Sharpe’s Corner. Situated between these two sites, the trestle itself is a potential green crab attractant. In other nearby bays like Samish and Padilla Bays, trappers have found concentrated green crab populations near structures like the trestle pillars provide.
In September 2022, Fidalgo Trestle team found a molt of a 22mm male green crab during their molt hunt shortly after a huge 700 trap, nine organization collaborative assessment effort turned up zero live green crabs, underscoring just how important complementary methods for green crab detections can be while local populations remain so low. To date, only two other green crab detections have been made in Fidalgo Bay, both by DNR efforts– a molt found in 2018 just north of the Trestle, and a live crab captured near Crandall Spit in August 2023 by staff with Washington Department of Natural Resources.
“Super” Volunteers
As is often the case, team members at the trestle site are deeply engaged in their marine stewardship community. Both Paul and Pete have been active members of the Skagit County Marine Resources Committee, and played a role in the growth of Crab Team during the early days. They obtained funding from the Northwest Straits Foundation to purchase additional gear and coordination coverage that supported Crab Team monitoring sites across the seven MRC counties. Team members also participate in other citizen science projects as “super volunteers,” including COASST, forage fish and salmon spawning surveys, and have been recognized for the breadth and depth of their contributions to volunteer stewardship.
“Crab Team is incredibly lucky to benefit from participation by community members who are truly leaders of marine conservation in their areas. Folks who have their fingers in many pots share the knowledge and connections they have across multiple projects. This is a powerful way that time and interest that is already donated to the effort goes even further to protecting and restoring shorelines.” reflects program lead, Emily Grason.
Olys on the Rise
Crab Team isn’t the only monitoring that happens under the trestle. Since 2002, Fidalgo Bay has been home to a long-term Olympia oyster (Ostrea lurida) restoration project, a collaboration of Skagit County Marine Resources Committee, Puget Sound Restoration Fund, Washington Department of Fish and Wildlife and others. What was initially an introduction of 50,000 hatchery seed oysters is now an established population of an estimated 5.5 million oysters as of 2023. It’s an example of an incredibly successful population enhancement.
A variety of monitoring methods have been used to assess the recovering populations of Olympia oysters. Bags full of oyster shells (“culch”) emptied at an annual timescale to count and measure all juvenile oysters who’ve taken up residence; stacks of glazed tiles checked twice monthly to count recently settled oyster larvae (“spat”); lowtide quadrat sampling to quantify oyster density and size. All of these efforts were supported by a substantial amount of hours donated by a pool of 21 dedicated volunteers over the last 5 years (and more spanning into the earliest days of the restoration).
The unique overlap in monitors between these two efforts adds a depth of experience that only sparks more questions for site captain Paul Dinnel, including, “will green crab prey on Olympia oysters [and] will the newly established oyster beds prove to be good habitat for green crabs?” Crab Team is particularly lucky to have Paul as a participant, because he has decades of research experience with Dungeness crab research and blue king crab, and knows more than almost anyone about how crabs use habitats in the region. His perspective brings an additional layer of context and expertise to monitoring in Fidalgo Bay. Thanks to the sustained efforts by Paul and other volunteer monitors on both topic areas, the questions about how green crabs could fit into the local picture may someday get answered.
–Lisa Watkins and Emily Grason
Header image caption: Monitors placed Fukui and minnow traps with bright orange bait jars for assessment trapping under the trestle in August 2022. Photo credit: Claire Cook
Photo Gallery
We love to get the virtual experience of monitoring with all of the Crab Team volunteers. Do you have a photo to share? Send it to crabteam@uw.edu. (Click on arrows to scroll, and photos to enlarge for more detail.)

























